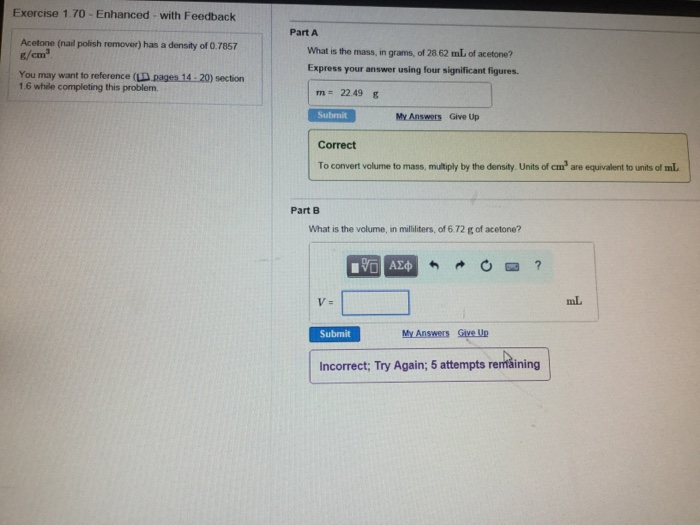
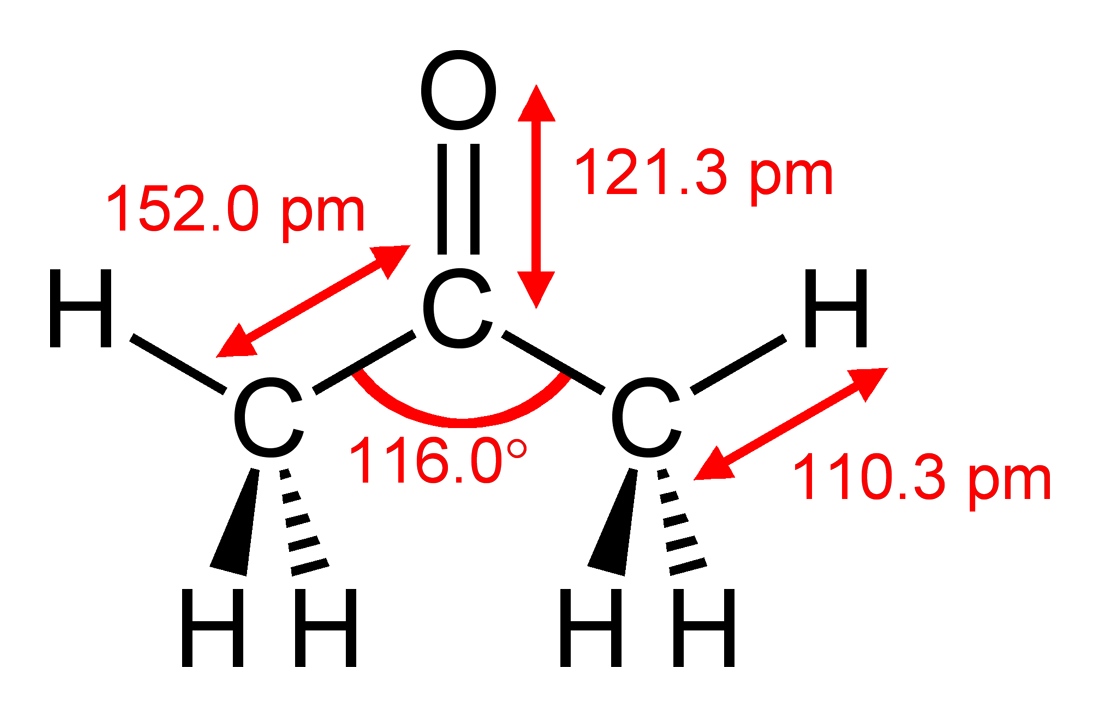
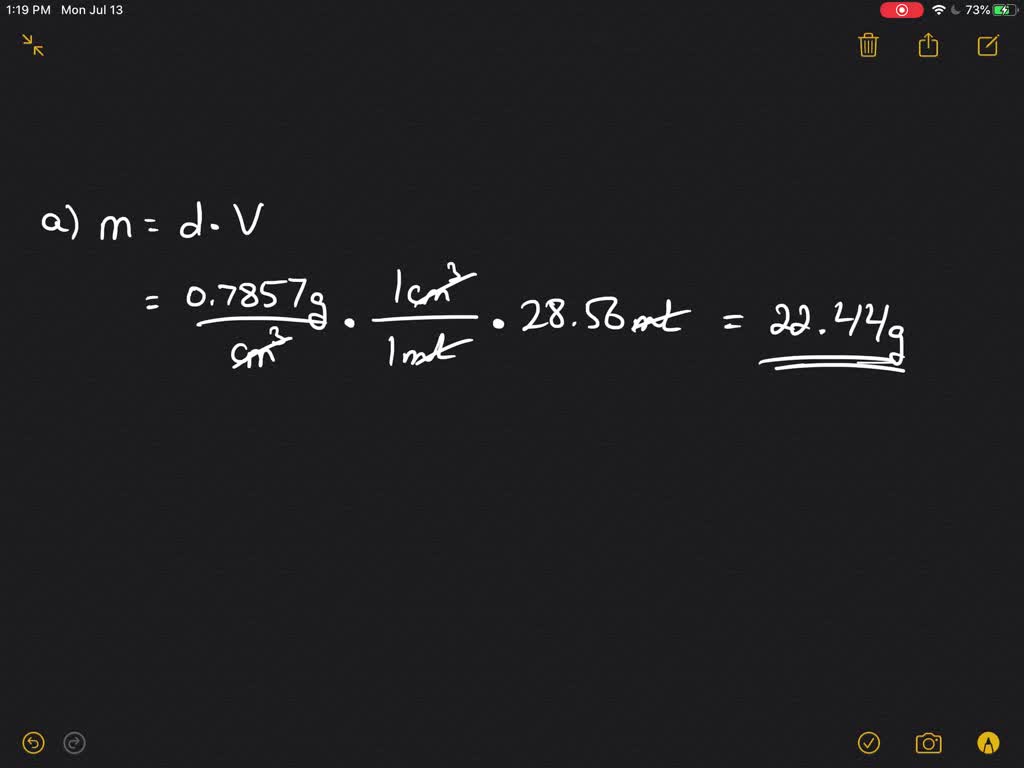✓ Solved: Calculate the molarity and mole fraction of acetone in a 1.00-m solution of acetone (CH3 COCH3)...

Competitive Adsorption of Methanol–Acetone on Surface Functionalization (−COOH, −OH, −NH2, and −SO3H): Grand Canonical Monte Carlo and Density Functional Theory Simulations | ACS Applied Materials & Interfaces

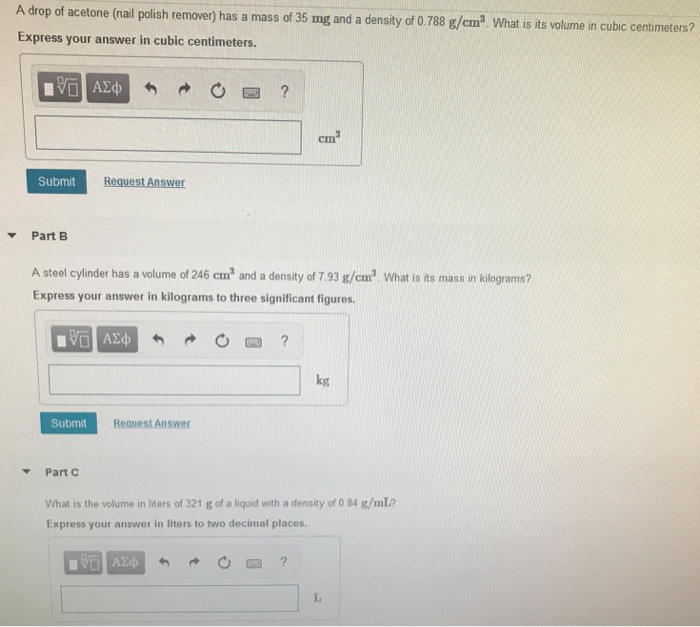
SOLVED:QUESTION 14 Acetone, C 3H 60, has a density of 0.788 g/cm 3 . A sample of acetone contains 2.7 x 10 24 molecules of acetone: Determine the volume; in mL, of

a) The penetration depth of some light and heavy ions inside acetone... | Download Scientific Diagram
An aqueous solution of acetone (MM = 58.08 g/mol) has a molality of 2.61 m and a density of 1.25 g/mL. What is the molarity of acetone in the solution? - Quora

Heavy Liquids Separation. Heavy Liquids Overview Methylene iodide (MI; ρ = 3.32 g/cm 3 ) is used to concentrate material according to density. When added. - ppt download

Highly Efficient Methyl Ketone Synthesis with Photoactivated Acetone and Olefins Assisted by Mg(II)-Exchanged Zeolite Y | The Journal of Organic Chemistry