Bevacizumab-awwb becomes first biosimilar approved for cancer treatment | MDedge Hematology and Oncology

Grpahical Abstract - The Benefits and Side Effects of Bevacizumab for the Treatment of Recurrent Ovarian Cancer

Randomized phase II trial of pemetrexed-cisplatin plus bevacizumab or thoracic radiotherapy followed by surgery for stage IIIA (N2) nonsquamous non–small cell lung cancer - The Journal of Thoracic and Cardiovascular Surgery

Bevacizumab and glioblastoma: Scientific review, newly reported updates, and ongoing controversies - Field - 2015 - Cancer - Wiley Online Library

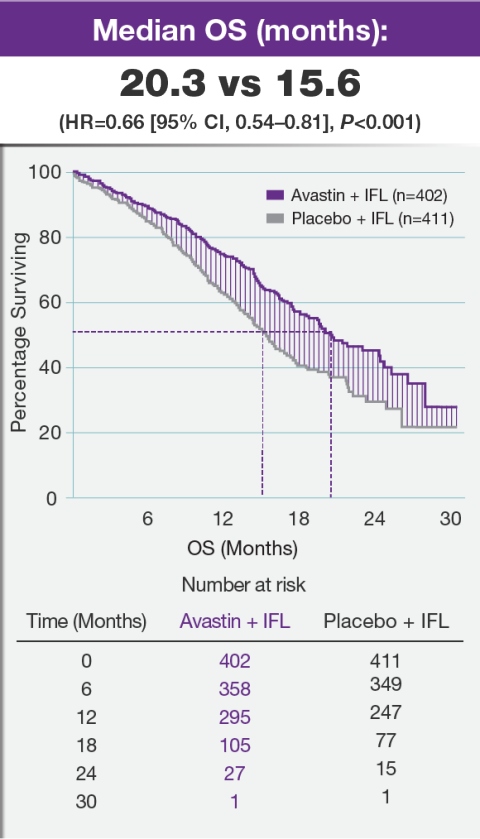
Bevacizumab (Avastin®) in cancer treatment: A review of 15 years of clinical experience and future outlook - Cancer Treatment Reviews

Efficacy of FOLFOXIRI plus bevacizumab in liver-limited metastatic colorectal cancer: A pooled analysis of clinical studies by Gruppo Oncologico del Nord Ovest - European Journal of Cancer

Nivolumab with carboplatin, paclitaxel, and bevacizumab for first-line treatment of advanced nonsquamous non-small-cell lung cancer - Annals of Oncology
Bevacizumab in Combination with Modified FOLFOX6 in Heavily Pretreated Patients with HER2/Neu-Negative Metastatic Breast Cancer: A Phase II Clinical Trial | PLOS ONE

Molecular targeted therapies in hepatocellular carcinoma: From pre-clinical models to clinical trials - Journal of Hepatology

Bevacizumab beyond first disease progression in metastatic colorectal cancer: a review of recent clinical trial data | Colorectal Cancer

FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial - The Lancet Oncology

FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial - The Lancet Oncology

Efficacy and Safety of Proposed Bevacizumab Biosimilar BE1040V in Patients With Metastatic Colorectal Cancer: A Phase III, Randomized, Double-blind, Noninferiority Clinical Trial - Clinical Therapeutics
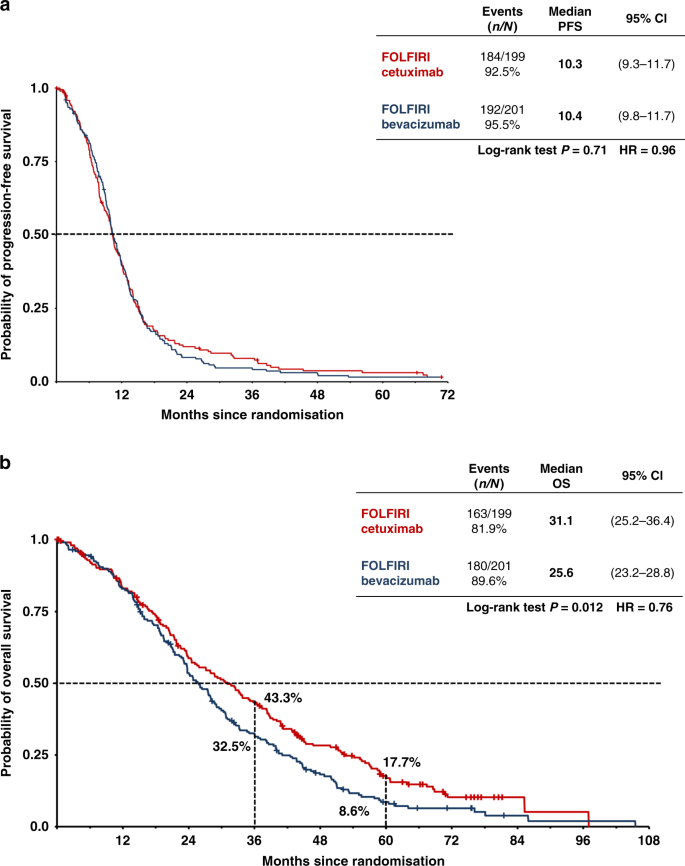
FOLFIRI plus cetuximab or bevacizumab for advanced colorectal cancer: final survival and per-protocol analysis of FIRE-3, a randomised clinical trial | British Journal of Cancer

Clinical and Regulatory Considerations for the Use of Bevacizumab Biosimilars in Metastatic Colorectal Cancer - ScienceDirect

A phase II pilot trial incorporating bevacizumab into dose-dense doxorubicin and cyclophosphamide followed by paclitaxel in patients with lymph node positive breast cancer: a trial coordinated by the Eastern Cooperative Oncology Group -

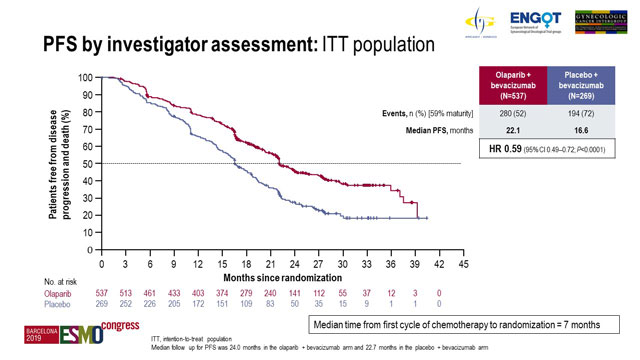
![PDF] Current and future directions of clinical trials for ovarian cancer. | Semantic Scholar PDF] Current and future directions of clinical trials for ovarian cancer. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7f15aa00fe070d53cbd782ae6153a6a322dec4f1/4-Figure2-1.png)