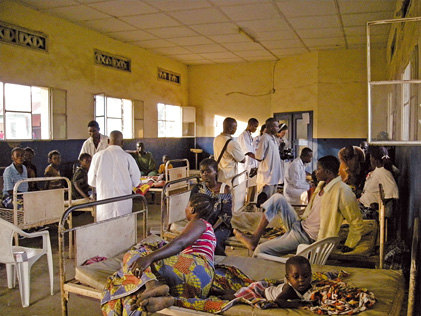
Prior Agreements in International Clinical Trials: Ensuring the Benefits of Research to Developing Countries
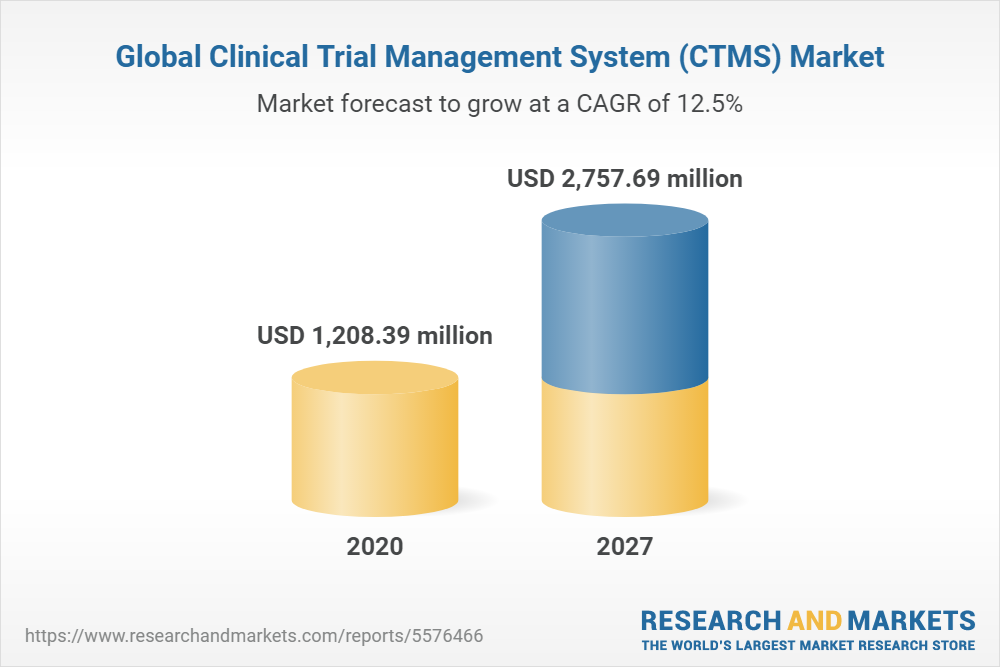
Global Clinical Trial Management System (CTMS) Market Report 2022: A $2.75+ Billion Market by 2027 - Increasing Innovative Technology Developments to Reduce the Rising Cost of Clinical Trials

Vaccine development and trials in low and lower-middle income countries: Key issues, advances and future opportunities. - Abstract - Europe PMC

The ethics of global clinical trials: In developing countries, participation in clinical trials is sometimes the only way to access medical treatment. What should be done to avoid exploitation of disadvantaged populations?:

Technopolis Group » Assessment of the performance and impact of the first programme of the European & Developing Countries Clinical Trials Partnership (EDCTP)
PLOS ONE: Participant perception, still a major challenge to clinical research in developing countries—A mixed methods study