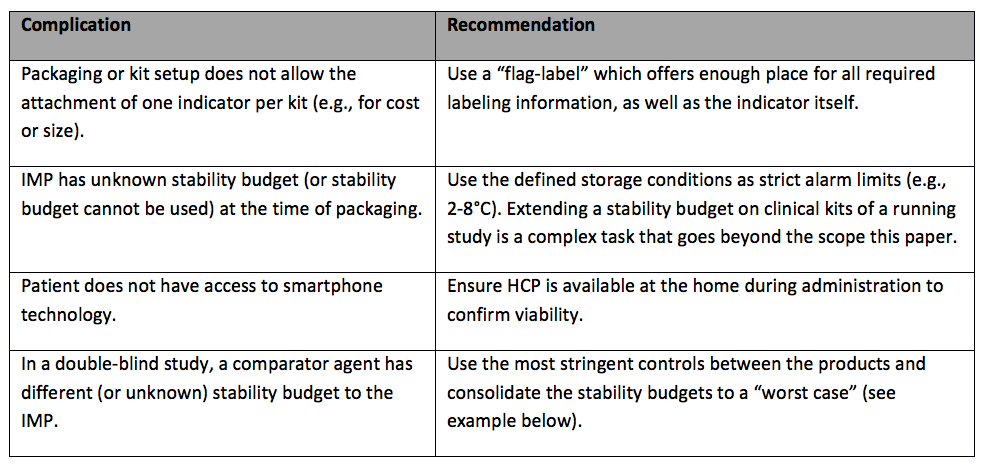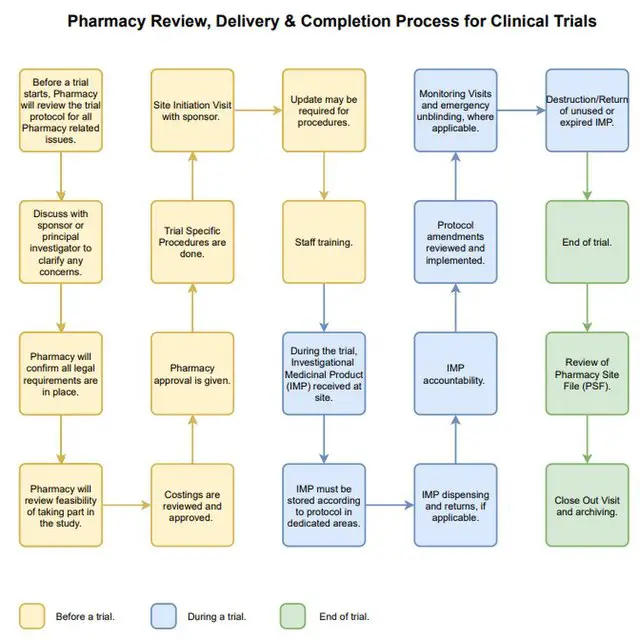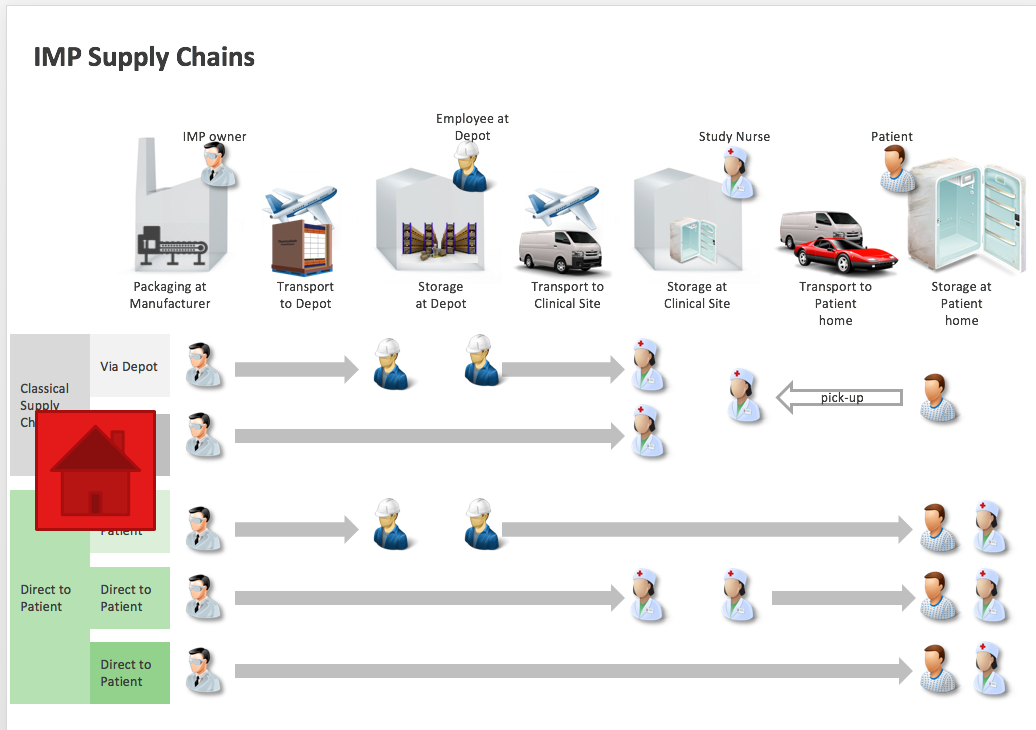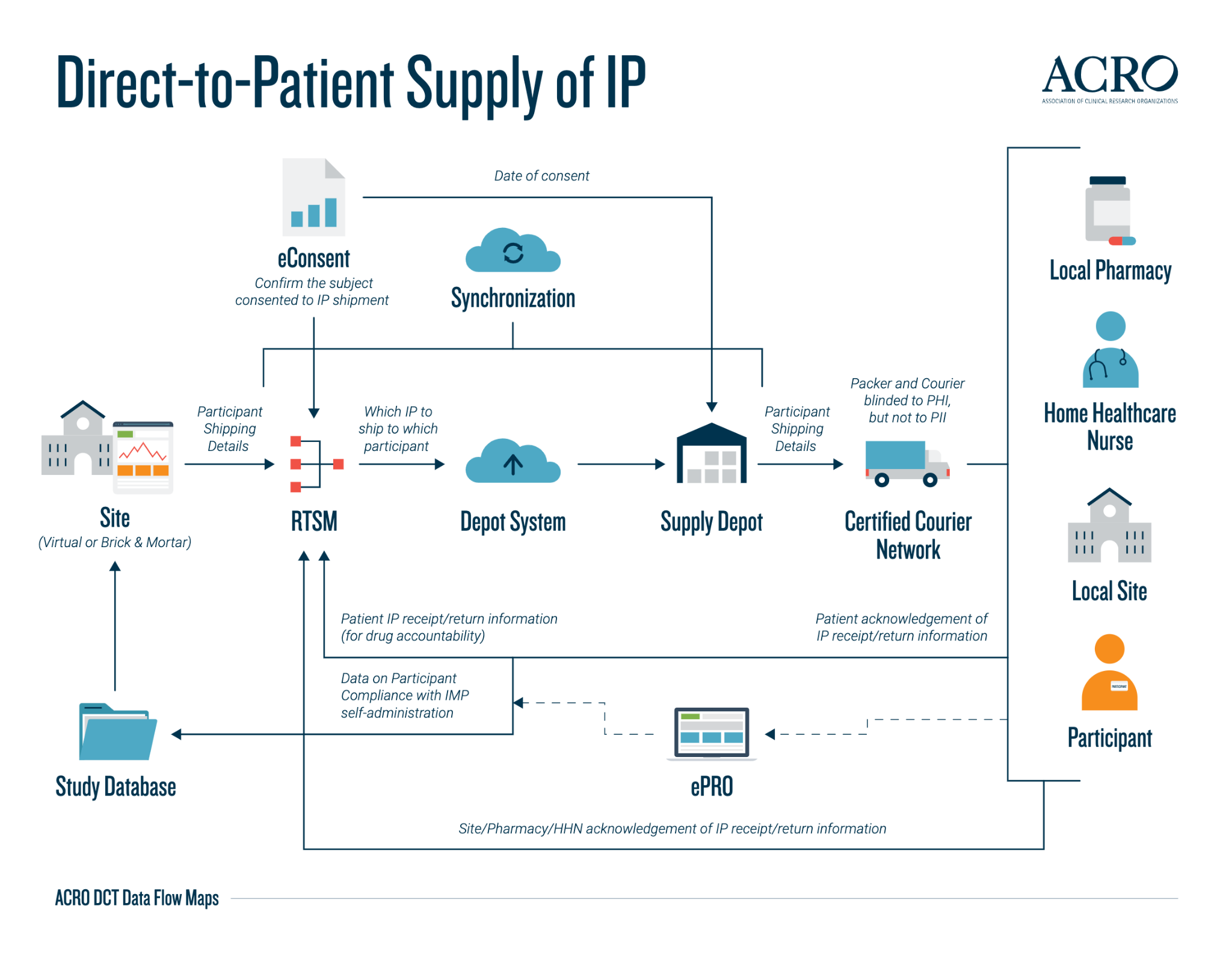
DCT(分散型臨床試験)データのフローマップ | Association of Clinical Research Organizations | Medidata Solutions - Medidata Solutions
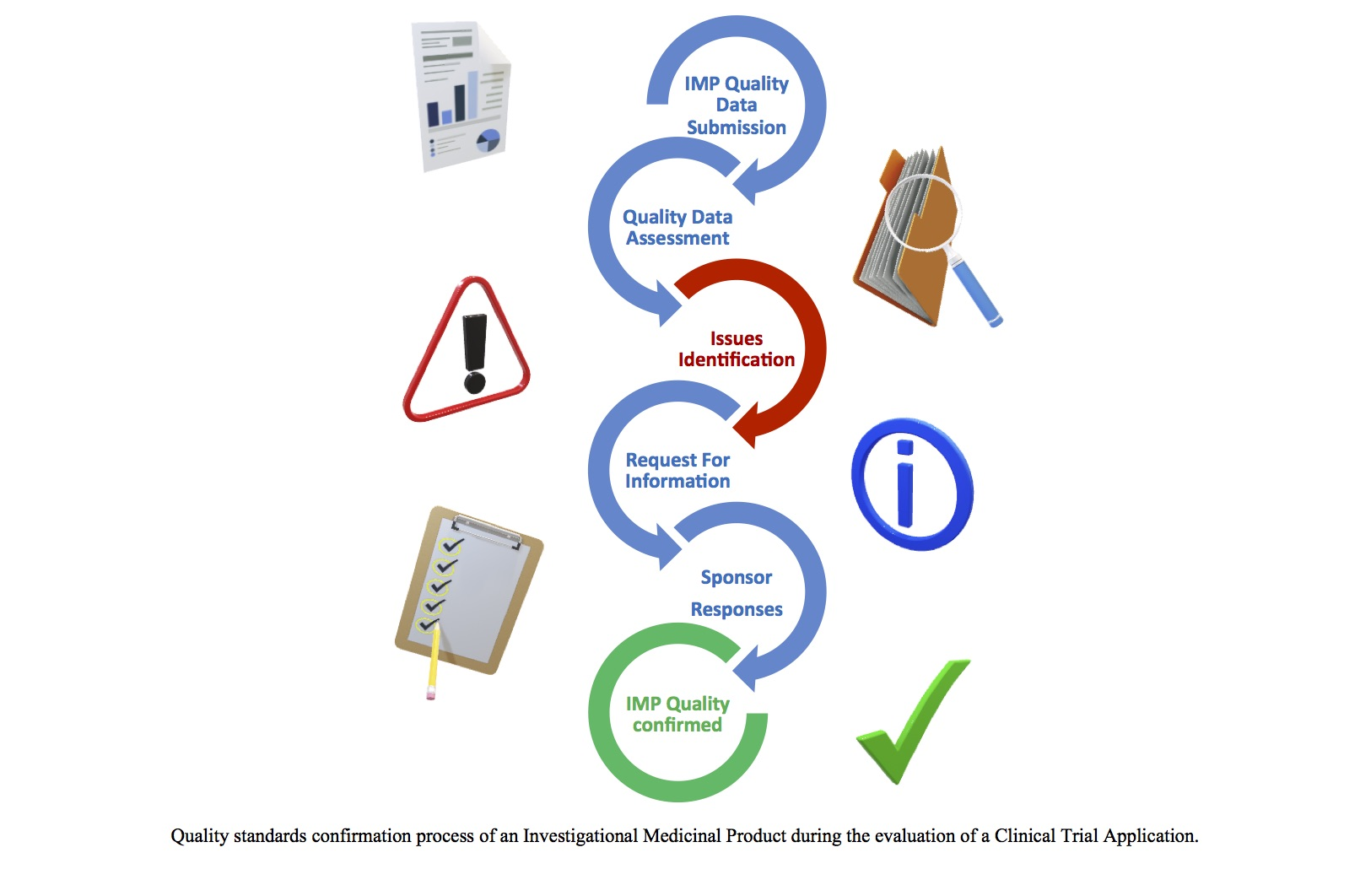
Pharmaceuticals | Free Full-Text | Quality Assessment of Investigational Medicinal Products in COVID-19 Clinical Trials: One Year of Activity at the Clinical Trials Office | HTML