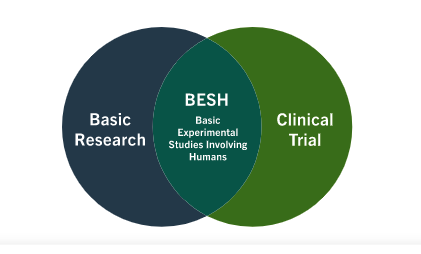
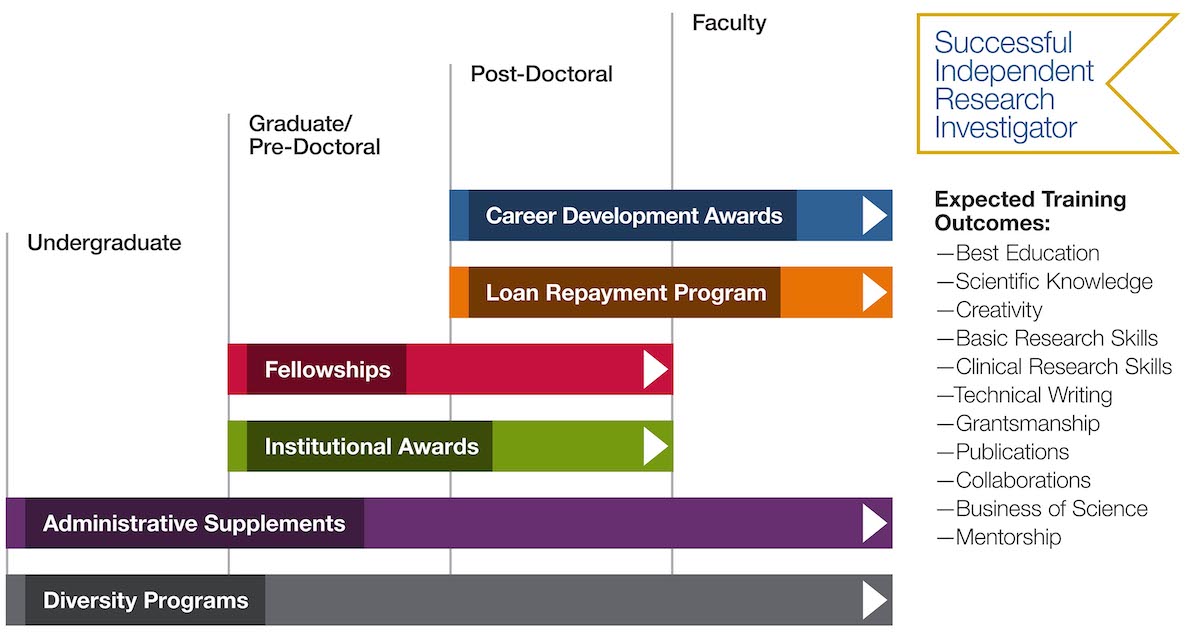
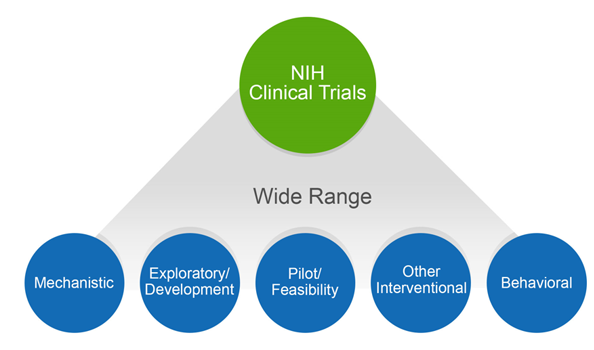
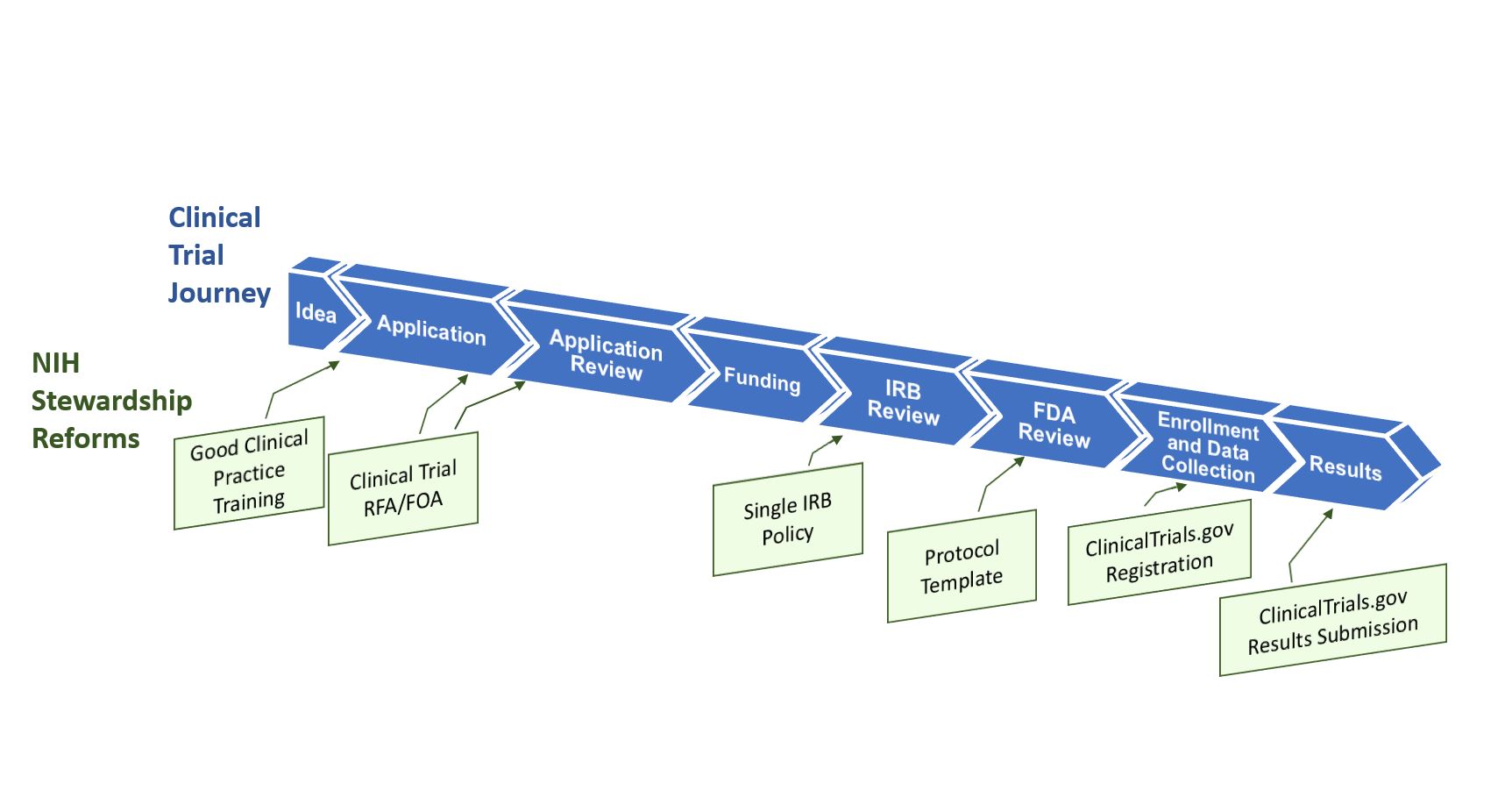
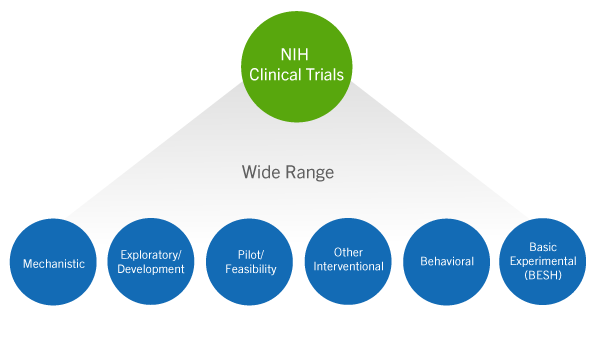
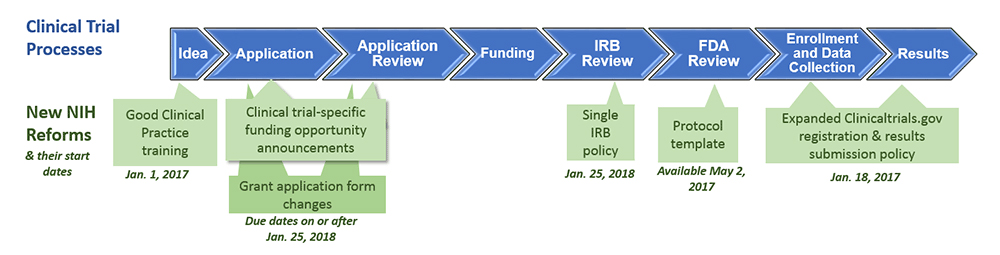
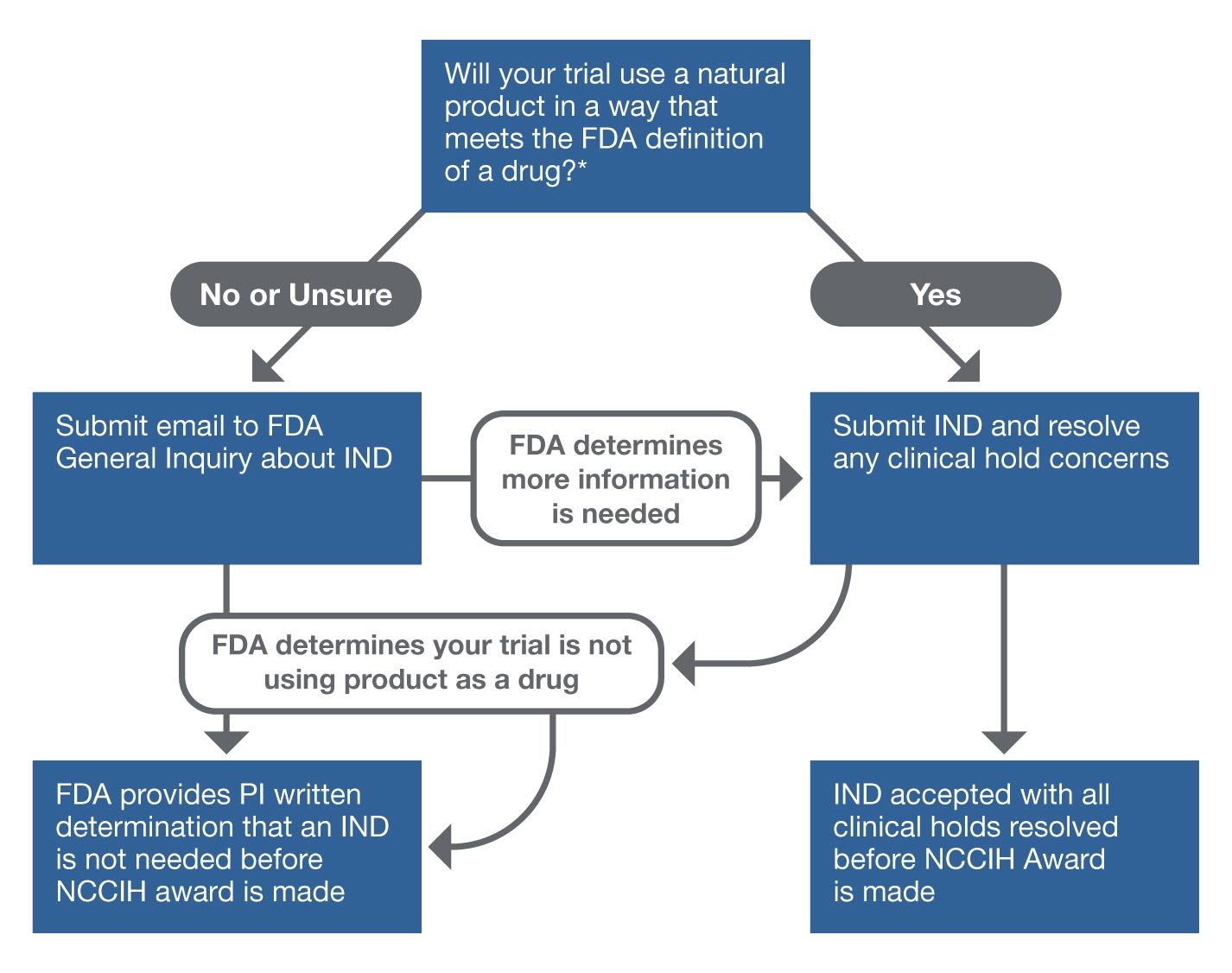
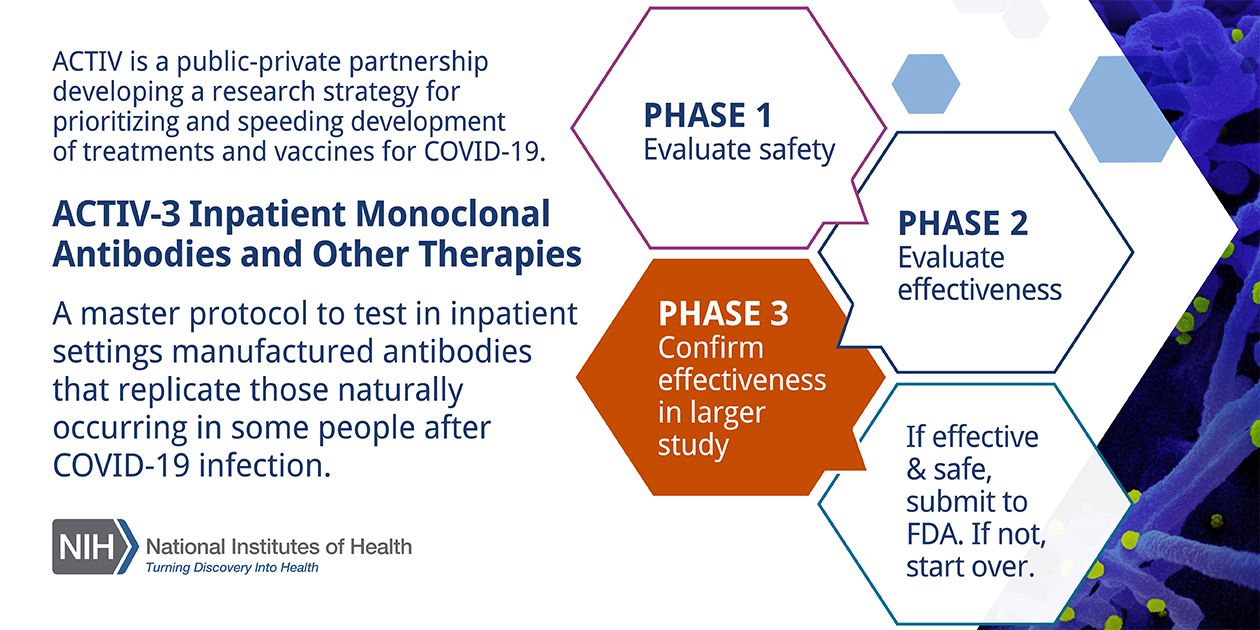
Free NIH clinical pharma and clinical research training sessions – Center for Bioethics and Research

Atlanta Pediatric Research | NIH Requirements for Human Subject Research | Clinical Research Resources | Research Resources | Research | Emory + Children's + GT | Atlanta Pediatric Research Alliance

Understanding The New NIH/HHS Final Rule On Clinical Trial Reporting – Tips For Compliance Success - Life Science Training Institute

Outcomes from the NIH Clinical Research Training Program: A Mentored Research Experience to Enhance Career Development of Clinician–Scientists | The Physician-Scientist Support Foundation

NIH-funded study to recruit thousands of participants to reveal exercise impact at the molecular level | National Institutes of Health (NIH)