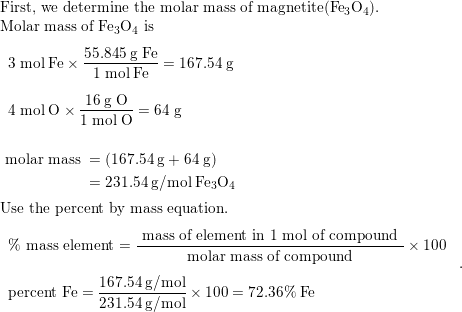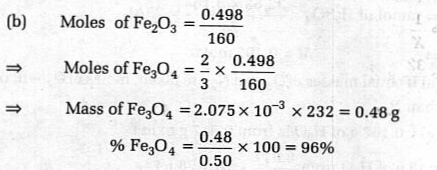
A 0.500 g sample of Magnetite ore (impure Fe_{3}O_{4}) istreated so that the iron is precipitated as Fe-IIIhydroxide. The precipitate is heated and converted to0.4980 g ee_{2}O_{3} . What is the percentage
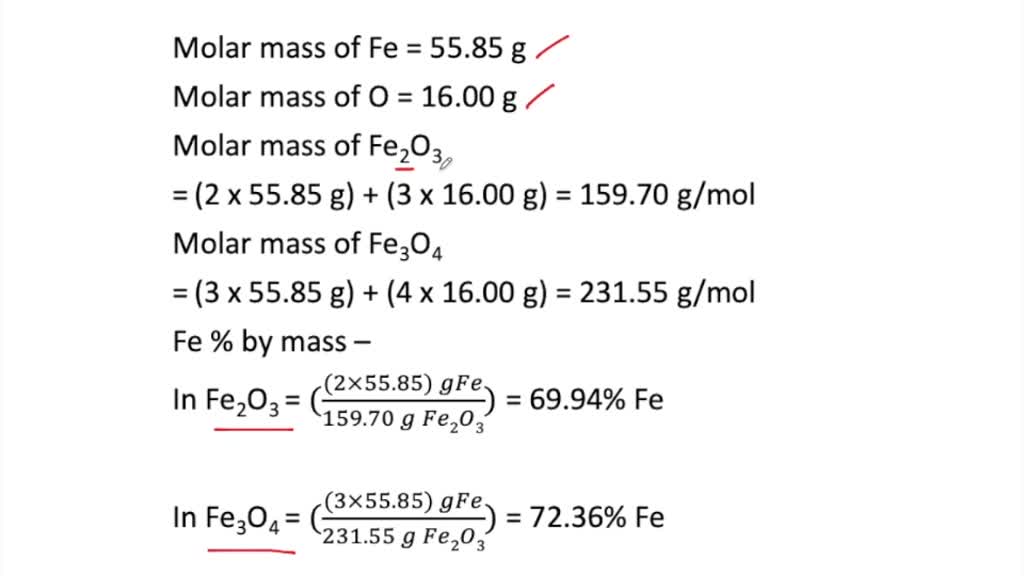
SOLVED:Analyze Hematite (Fe \mathrm{O}_{3} ) and magnetite \left(\mathrm{Fe}_{3} \mathrm{O}_{4}\right) are two ores used as sources of iron. Which ore provides the greater percent of iron per kilogram?

SOLVED:Magnetite is 4 binary compound containing only iron (55.5 gmol) and oxygen (16 g/mol) . The weight percent of iron is 72.36% . What is the empirical formula magnetite? FeO FeOz C.

Magnetic characterisation of magnetite and hematite from the Blötberget apatite – iron oxide deposits (Bergslagen), south-central Sweden
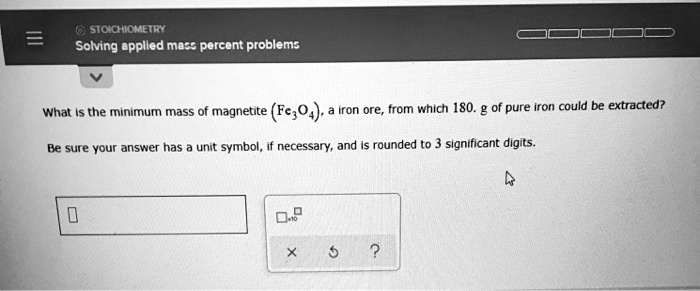
SOLVED:0 STOICHIOMETRY Solving opplled _ Mdbe percent problem: What the minimum mass of magnetite (Fe;04) Iron ore from which 180. g of pure Iron could be extracted? Be sure your answer has
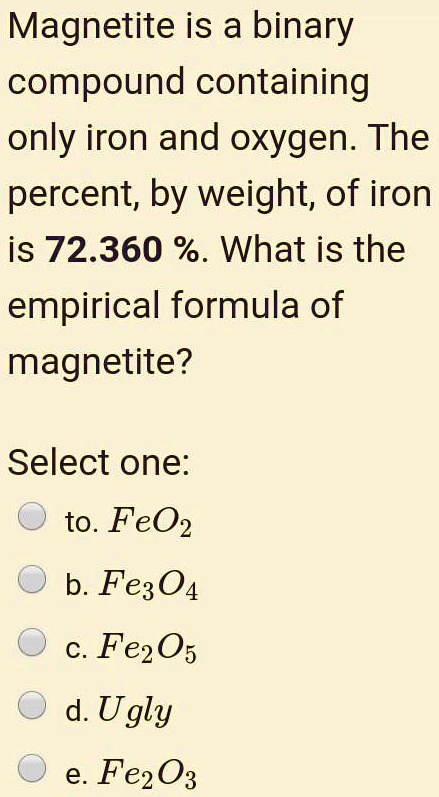
SOLVED:Magnetite is a binary compound containing only iron and oxygen: The percent; by weight; of iron is 72.360 %. What is the empirical formula of magnetite? Select one: to. FeO2 b. Fe304