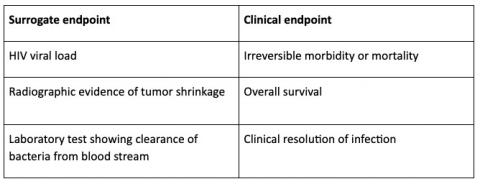
Time to Review the Role of Surrogate End Points in Health Policy: State of the Art and the Way Forward - Value in Health
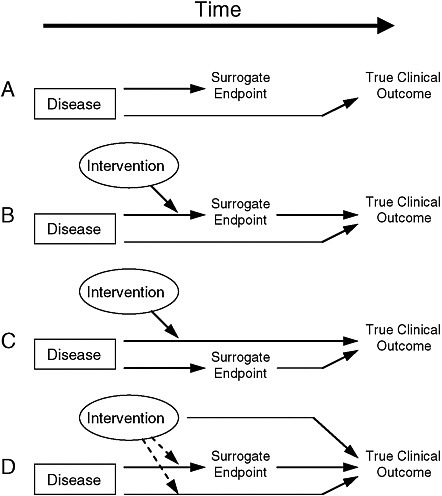
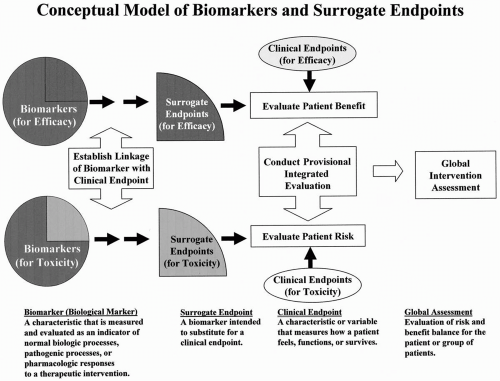
2 Review: Evaluating and Regulating Biomarker Use | Evaluation of Biomarkers and Surrogate Endpoints in Chronic Disease |The National Academies Press
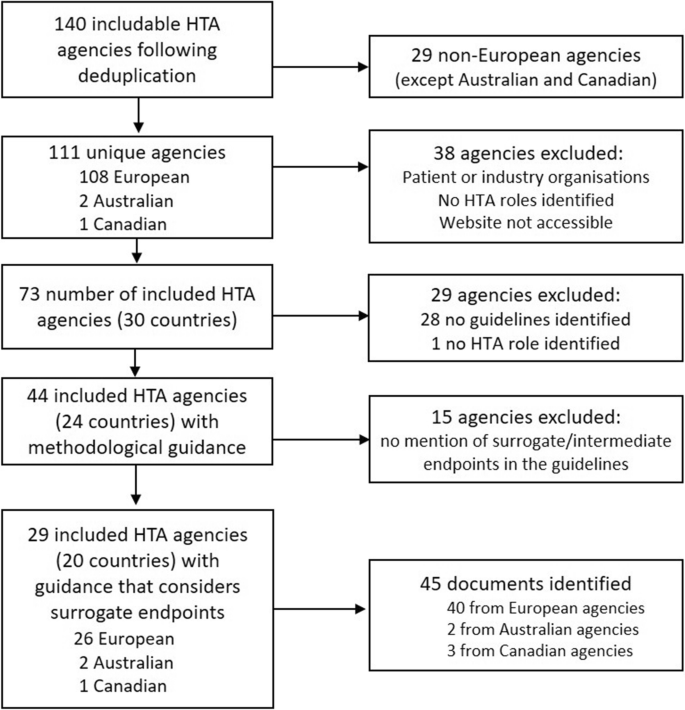
Surrogate Endpoints in Health Technology Assessment: An International Review of Methodological Guidelines | SpringerLink

The role of imperfect surrogate endpoint information in drug approval and reimbursement decisions - ScienceDirect

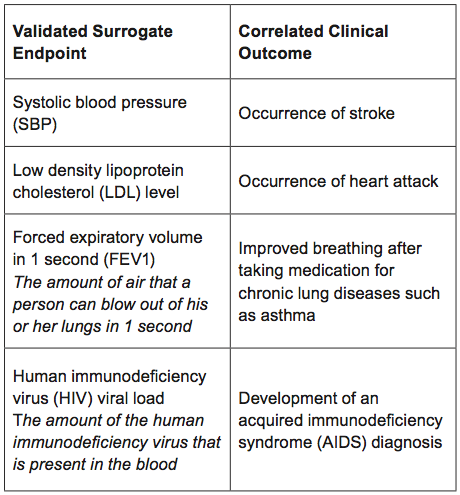
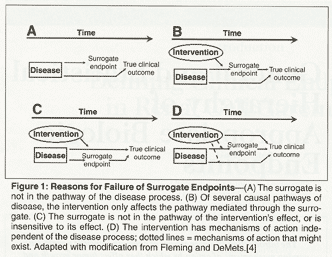
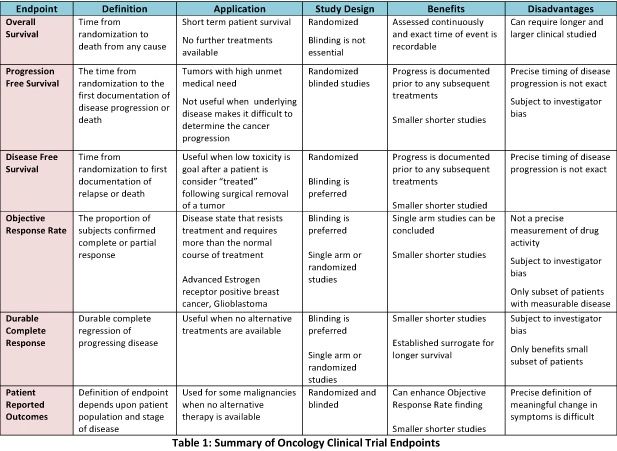
Surrogate Endpoints in Infectious Diseases Trials: FDA Perspective Surrogate Endpoints in Infectious Diseases Trials: FDA Perspective John H. Powers, MD. - ppt download
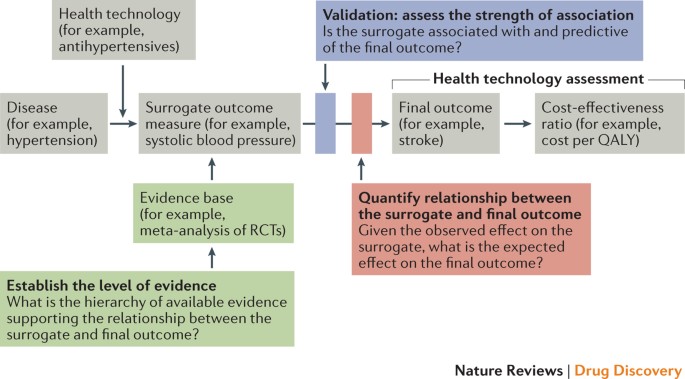
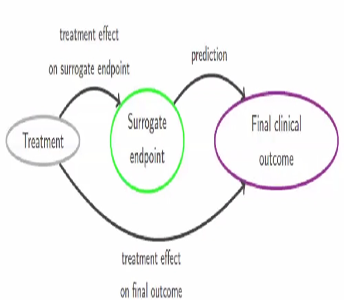
Use of surrogate end points in healthcare policy: a proposal for adoption of a validation framework | Nature Reviews Drug Discovery